New physics-based computation and AI framework at U-M explores aggressive behavior of cancer cells
Media contact: Eric Shaw, 734-647-1970 | Patients may contact Cancer AnswerLine™, 800-865-1125
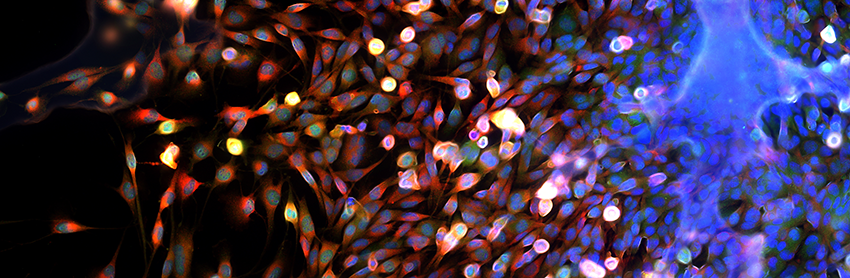
A team of interdisciplinary researchers at the University of Michigan, backed by a $1 million W.M. Keck Foundation grant, has developed a high-risk, high-reward approach to understand how each cell in a population processes information and translates that to action driving cancer cell progression.
Cancer is an illness caused by an uncontrolled division of transformed cells, which can originate in almost any organ of the body. Tremendous variability exists in the progression of disease and response to therapy among different people with the same general type of cancer, such as breast cancer. Even at the level of a single person, cancer cells show tremendous heterogeneity within a single tumor and among a primary tumor and metastases. This heterogeneity causes drug resistance and fatal disease.
“The prevailing dogma is that heterogeneity among cancer cells arises randomly, generating greedy individual cancer cells that compete for growth factors and optimal environments. The rare 'winners' in this competition survive and metastasize,” said Gary Luker, M.D.,, professor of radiology, biomedical engineering, microbiology and immunology. “However, we often see tumors that consistently maintain heterogeneous subpopulations of cancer cells, some of which appear less able to grow and spread.”
This observation prompted Luker and research colleague Kathryn Luker, Ph.D., associate research scientist in radiology, to hypothesize that cancer cells may actually collaborate under some circumstances to cause disease and not just to compete.
The Lukers shared their hypothesis with U-M experts in computational science and artificial intelligence during a Michigan Institute for Computational Discovery and Engineering (MICDE) faculty workshop, which brought together an interdisciplinary group of researchers to identify ways to advance artificial intelligence and machine learning methods for biomedical problems.
A subset of these researchers, including Jennifer Linderman, Ph.D., professor of chemical and biomedical engineering, was awarded an MICDE catalyst grant and a Michigan Institute for Data Science (MIDAS) PODS grant, which they used to establish the proof of concept and to generate preliminary results. MICDE and MIDAS are based in the Office of the Vice President for Research.
“Advancements in computational science allow us to acquire and analyze tremendous amounts of data, to solve some of today’s most pressing challenges, considered to be previously unattainable,” said MICDE Director Krishna Garikipati, Ph.D., professor of mathematics and mechanical engineering. “Discovering how cells communicate and drive the progression of cancer, to a large degree, is possible because the University of Michigan research environment encourages an interdisciplinary approach to unleash the potential of computational discovery.”
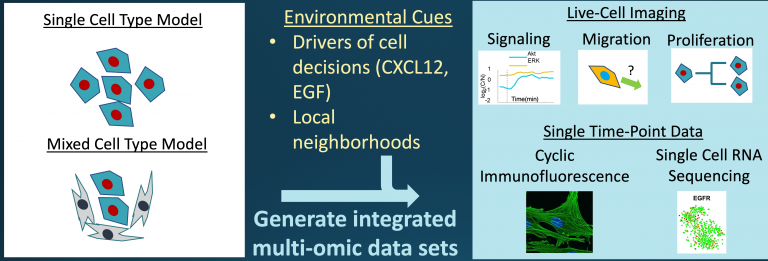
graphic courtesy of U-M Office of Research
Building on large, single-cell datasets unique to the team, researchers combine inverse reinforcement learning, an artificial intelligence method typically applied to discover motivations for human behaviors, with computational models inferred based on the physics and chemistry of cell signaling and migration.
This research proposes an entirely new conceptual approach combining single-cell data, physics-based modeling and artificial intelligence to single-cell heterogeneity and intercellular interactions.
“By discovering testable molecular processes underlying ‘decision-making’ by single cells and their ‘motivations’ for acting competitively or collaboratively, this research blazes a new path to understand and treat cancer,” said Kathryn Luker. “The idea that single, heterogeneous cancer cells work collectively within a constrained range of variability to drive population-level outputs in tumor progression is a ground-breaking concept that may revolutionize how we approach cancer biology and therapy.”

image courtesy of U-M Office of Research
The W. M. Keck Foundation was established in 1954 in Los Angeles by William Myron Keck, founder of The Superior Oil Company. One of the nation's largest philanthropic organizations, the W. M. Keck Foundation supports outstanding science, engineering and medical research.
“While the current proposal focuses on cancer, this innovative computational framework represents a transformative leap with widespread applications in multiple other biomedical, physical, and social sciences,” added Garikipati. “MICDE will continue to support innovative and interdisciplinary projects aiming to advance the current paradigms.”
Learn more about this project: https://micde.umich.edu/feature/computational-framework-and-cancer-keck-foundation/
